External Sulfate Attack
Concrete Experts International has extensive, world-wide experience with deteriorated concrete suffering from varies types of sulfate attack caused by seawater, ground water, aggressive water from sewer systems and fresh water. Diagnosing external sulfate attack is an integrated part of our petrographic analysis of concrete.
Other
Deterioration
Mechanisms
External Sulfate Attack
Concrete Experts International has extensive, world-wide experience with deteriorated concrete suffering from sulfate attack caused by water containing sulfate. Diagnosing external sulfate attack is an integrated part of our petrographic analysis of concrete
What is External Sulfate Attack?
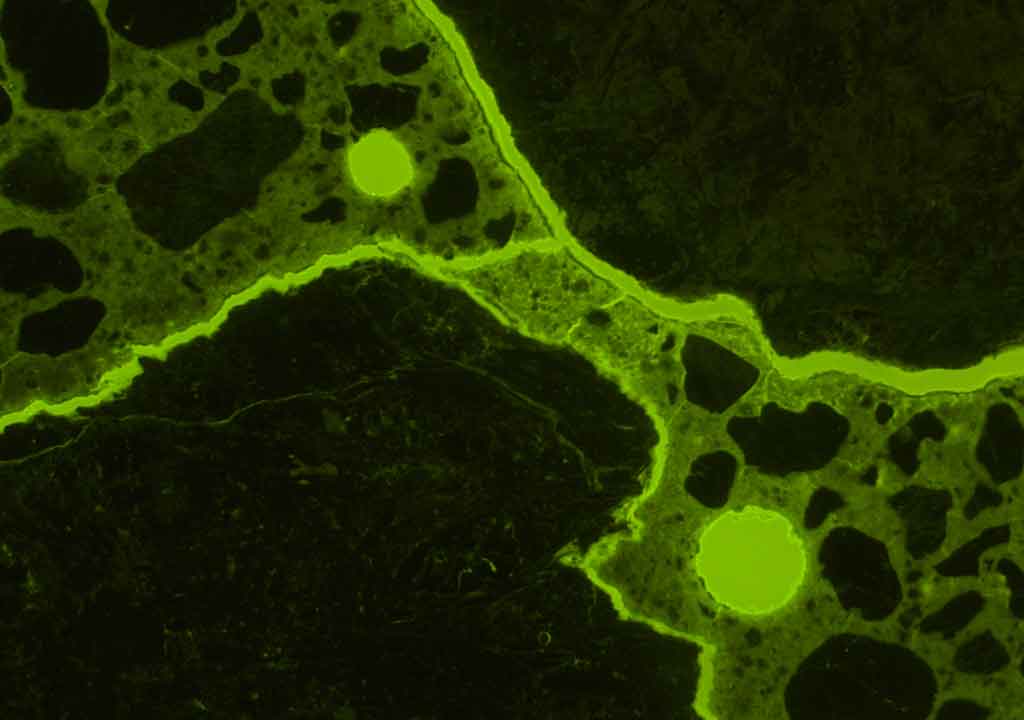
External sulfate attack is a chemical breakdown mechanism where sulfate ions from an external source attack components of the cement paste. Such attack can occur when concrete is in contact with sulfate containing water e.g. seawater, swamp water, ground water or sewage water. The often massive formation of gypsum and ettringite formed during the external sulfate attack may cause concrete to crack and scale. However, both laboratory studies and examinations of field concrete show that external sulfate attack is often manifested, not by expansion or cracking, but by loss of cohesion and strength.
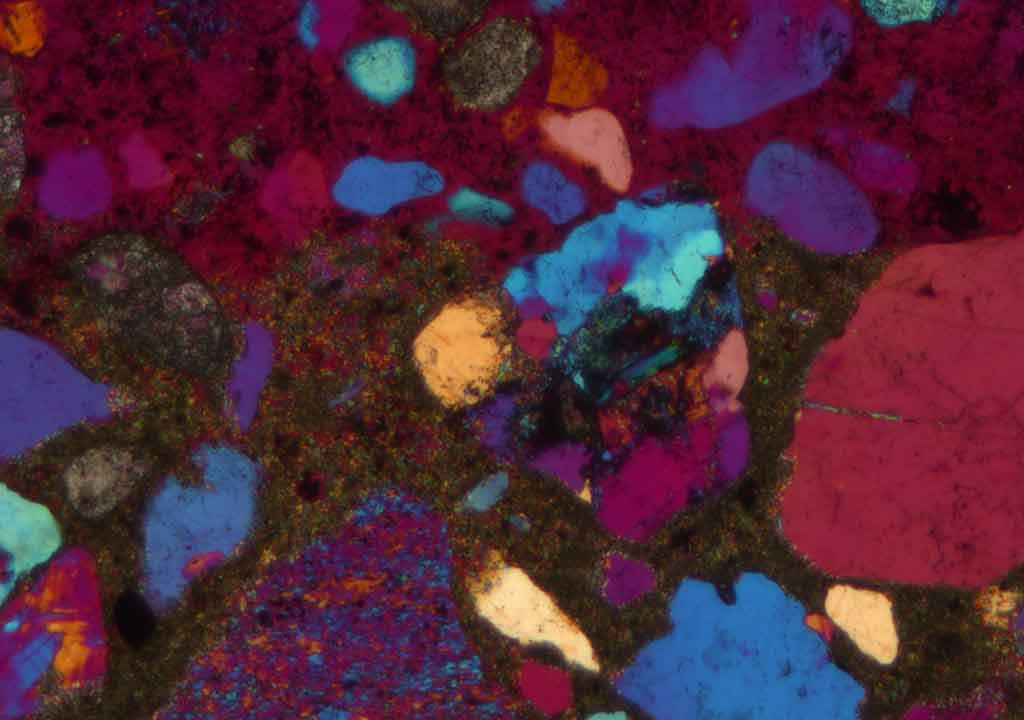
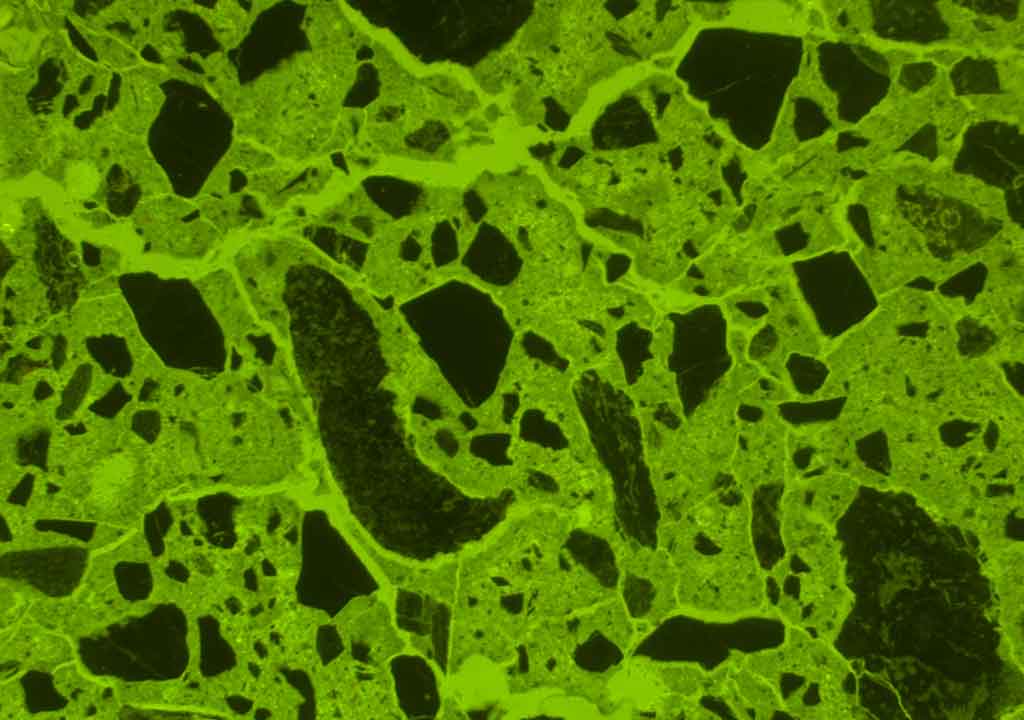
Microscopic appearance
The microscopic appearance of concrete suffering from external sulfate attack appears to be quite variable. Some diagnostic features such as
- Surface parallel cracks
- Presence of gypsum and ettringite
- Depletion of calcium hydroxide, and
- Decalcification of C-S-H
- Popcorn carbonation
are, however, often found associated with external sulfate attack.
The above-mentioned features are usually most pronounced near the attacked surface. Sometimes external sulfate attack causes the paste to expand and gaps around aggregates are formed. All the features do not necessarily have to be observed to diagnose an external sulfate attack. The features present depend on many factors such as the quality of the concrete (including w/c and cement type), exposure time, temperature, concentration, and chemistry of the ambient water.
There is a general agreement that concrete suffering from external sulfate attack develops a more and less pronounced mineralogical and chemical zoning which can be studied in the optical fluorescence microscope and the scanning electron microscope.
ASTM C856 recommends chemical analysis to verify that the sulfate content of the concrete has been increased over that expected from the concrete constituents in order to diagnose external sulfate attack.
Please to not hesitate to contact CXI if you have some problems regarding external sulfate attack or any other deterioration mechanisms.
Terms and definitions
Using the same terms makes communication better! We have made this small dictionary which describes some of the terms used in concrete examination and description.
Terms and Definitions
The following small dictionary describes some of the terms used in concrete examination and description.
Agglomerate: 3 or more air voids in close contact
Aggregate: Component of the concrete consisting of a coarse aggregate fraction (more than 4 mm) and a fine aggregate fraction (less than 4 mm)
Alite: Tricalciumsilicate, C3S (3CaO-SiO2), a natural occurring mineral present in ordinary Portland cement.
Alkali silica reaction: Reaction between silica in the aggregates (e.g. chert, flint, micro-crystalline quartz), alkali (from cement, de-icing salt, sea water), calcium hydroxide and water. Alkali silica reaction causes aggregates to expand and cracks are formed radiating out into the ambient cement paste.
Alkali silica gel: A product of alkali silica reaction. Gel is usually clear, transparent and non-crystalline (amorphous), although in some cases it may crystallize. It expands during water absorption. Gel is usually found lining or filling cracks and air voids.
Aluminate phase: Tricalciumaluminate, C3A (3CaO-Al2O3)
Anhydrate: CaSO4
Belite: Dicalciumsilicate, C2S (2CaO-SiO2), natural occurring mineral present in ordinary Portland cement.
Bleeding: Water separation in the concrete. "Bleeding" is the phenomenon where water migrates towards the surface of the concrete and collects on the surface or underneath coarse aggregates. Seen as narrow voids along the interface between aggregate and cement paste. Calcium hydroxide may be deposited in the voids.
Calcium carbonate: CaCO3 , calcite, found as deposits in voids and cracks and on the surface.
Calcium hydroxide: Ca(OH)2, Portlandite, hydration product of C3S and C2S. Appears throughout the non-carbonated cement paste and occasionally as larger crystals in voids and cracks
Carbonation: Transformation of the calcium containing constituents of the concrete by reaction with the carbon dioxide in air. Calcium hydroxide is transformed to calcium carbonate and consequently the paste becomes chemically neutral.
Cement: The visible cement minerals are: C3S, C2S and C4AF. C3A is not visible.
Cement paste: Compound of hydration products from the cement-water reaction and unhydrated cement. Micro silica (MS) and fly ash (FA) is usually treated as part of the paste.
Cracks: Are divided in 3 groups in relation their width: coarse cracks are cracks wider than 0.1 mm, fine cracks varies from 0.01 - 0.1 mm and micro cracks are less than 0.01 mm. Cracks usually run in the cement paste; however, occasionally the aggregates are traversed. Cracks form either in the early (plastic stage) or at a later stage (fully hardened stage) in the life of the concrete .
Delayed Ettringite Formation (DEF): Hardened concrete that has been subjected to high-temperature heat curing or high internal hydration heat temperature may suffer from expansion and cracking during subsequent exposure to moisture. DEF is diagnosed by the presence of gaps around aggregate particles.
Entrained air: Usually defined as the small and medium sized spherical air voids with maximum size 0.5 - 1 mm
Entrapped air: Comprises irregular and angular air voids of all sizes
Ettringite: 3CaO.Al2O3.3CaSO4.32H2O. Needle shaped crystals of calcium sulfo-aluminate hydrate produced by constituents in the cement paste, including gypsum. It is also produced by sulfate attack on the concrete.
Ferrite phase: Tetracalciumaluminateferrite, C4AF (4CaO-Al2O3-Fe2O3)
Filled voids: Air voids with complete or partial filling of e.g. alkali silica gel, ettringite, calcium hydroxide, calcium carbonate or gypsum.
Friedel Salt (mono-chloride): 3CaO.Al2O3.CaCl2.10H2O
Gaps: Cracks observed all the way around aggregate particles in the interface zone between aggregate and cement paste. Gaps can be empty, or be partly or completely filled with ettringite. The width of the gaps is positively correlated with the size of the aggregate. Gaps are evidence of some sort of paste expansion occurring from delayed ettringite formation (DEF), freeze/thaw and sulfate attack.
Gypsum: CaSO4.2H2O, calcium sulfate which is found as a common constituent of cement. It is also produced by acid attack (sulfuric acid) on concrete.
Hydration: Reaction between cement and water, producing cement gel (cement paste, cement glue) serving as an adhesive in the concrete.
Mono-sulfate: 3CaO.Al2O3.CaSO4.12H2O
Plastic shrinkage: Shrinkage caused by strong drying of newly cast concrete. The result is often plastic shrinkage cracks in the concrete surface.
Plastic settlement: Settlement of fresh concrete, often causing plastic settlement cracks.
Re-crystallization: Dissolution of crystals and re-deposition either of the same mineral in a different form or of new minerals, mostly stabilized due to the presence of water.
Segregation: "Segregation" is the phenomenon where the concrete has no internal adhesion, and the mortar (therefore separates from the coarse aggregates.
Thaumasite: CaSiO3.CaSO4.CaCO3. A mineral not naturally present in concrete. Thaumasite may occur in concrete suffering from sulfate attack. The presence of thaumasite depends according to literature on temperature and presence of carbon dioxide.
w/c ratio: The ratio of water to cement by weight of the cement paste. If the cement paste contains fly ash (FA) or micro silica (MS) the addition of these materials can be taken into account using an activity factor and an equivalent w/c ratio can be calculated.
Other Deterioration Mechanisms
Contact Us
Concrete Experts International ApS
Gøngehusvej 242
DK-2950 Vedbæk